Welcome back! In the last post, we explored how hydrocarbons relate through the FID mole response factor, and hopefully you’ve already tried applying it. That’s probably why you’re here again — ready to look at the same idea but in terms of the FID mass response factor. I appreciate you stopping by, and I’m excited to walk through this next part of the series with you. Just before we start, make sure you’ve understood what we covered in the previous article about FID Mole Response Factor because we’re building directly on top of that logic.
Alright, enough chit-chat. Let’s dive into how the FID mass response factor works.
How the FID Mass Response Factor Works
First, let’s tackle the FID mass response factor. When we talk about mass, the units could be mass%, ppm mass, mg/kg, mg/L, and so on. Just like before, we return to the combustion reaction inside the FID flame:
CxHy + O2 → xCHO+ + e−
To keep the explanation consistent with the previous post, we’ll use Methane and Propane again. Imagine a mixture containing “x” mass% Methane and “x” mass% Propane. Now we need to convert those mass-based values into something mole-related, and to do that, we use molecular weight (MW):
- “x” mass% Methane → x / 16 mole-related Methane
- “x” mass% Propane → x / 44 mole-related Propane
Now that both are mole-related, we can apply the same logic as before:
x/16 mole-related Methane → “z” FID peak area
Which means:
x/44 mole-related Methane → (z * x/44) / (x/16) FID peak area
Or more simply:
x/44 mole-related Methane → 16z / 44 FID peak area
Because Propane produces three times the CHO⁺ ions per mole-unit, we get:
x/44 mole-related Propane → 3 * 16z / 44 FID peak area
Now that we know the peak area for each, let’s compute the FID mass response factor:
RF(Methane) = x / z
RF(Propane) = x / (3 * 16z / 44)
Finally, the relative response factor (RRF) by mass — keeping Methane as the reference:
RRF(Propane/Methane) = (x / (3*16z/44)) / (x / z)
Solving the expression:
RRF(Propane/Methane) = 1 / (3*16/44)
Or expressed differently:
RRF(Propane/Methane) = 44 / (3*16)
Now that we recognize those numbers —
44 (MW of Propane),
3 (carbon ratio Propane : Methane),
16 (MW of Methane) —
we can finally generalize:
RRF(i/ref) = MW_i / ((C_i / C_ref) * MW_ref)
or equivalently:
RRF(i/ref) = (C_ref * MW_i) / (C_i * MW_ref)
It took a few steps to reach the final equations, but the goal is to help you understand how everything comes together. Once you grasp the logic, you can simply use the final formula whenever needed — and re-derive it confidently if required.
Examples of the FID Mass Response Factor
Let’s work through a quick exercise. Try calculating the FID mass response factor for Pentane versus Ethane (Ethane as reference) before checking the answer.
RRF(Pentane/Ethane) = (2 * 72) / (5 * 30) = 0.96
Another one — Heptane against Ethane:
RRF(Heptane/Ethane) = (2 * 86) / (7 * 30) = 0.82
Now that you understand how to calculate it, let’s plug the formula into Excel and compute several values. You’ll soon see why this matters.
RRF by Mass Table (Pentane as Reference)
| Compound | Carbon | MW | RRF-mass (Pentane Ref) |
|---|---|---|---|
| Methane | 1 | 16 | 1.111 |
| Ethane | 2 | 30 | 1.042 |
| Propane | 3 | 44 | 1.019 |
| Butane | 4 | 58 | 1.007 |
| Pentane | 5 | 72 | 1.000 |
| Hexane | 6 | 86 | 0.995 |
| Heptane | 7 | 100 | 0.992 |
| Octane | 8 | 114 | 0.990 |
| Nonane | 9 | 128 | 0.988 |
| Decane | 10 | 142 | 0.986 |
| Undecane | 11 | 156 | 0.985 |
| Dodecane | 12 | 170 | 0.984 |
| Tridecane | 13 | 184 | 0.983 |
| Tetradecane | 14 | 198 | 0.982 |
| Pentadecane | 15 | 212 | 0.981 |
| Hexadecane | 16 | 226 | 0.981 |
| Heptadecane | 17 | 240 | 0.980 |
| Octadecane | 18 | 254 | 0.980 |
| Nonadecane | 19 | 268 | 0.980 |
| Eicosane | 20 | 282 | 0.979 |
Notice how the values change very little? That means their response factors are very similar — and that’s exactly why international methods like ASTM D2887 and ASTM D6352 allow applying the same response factor across the whole chromatogram. Now you know the science behind it.
Comparing Experimental and Theoretical RRF by Mass
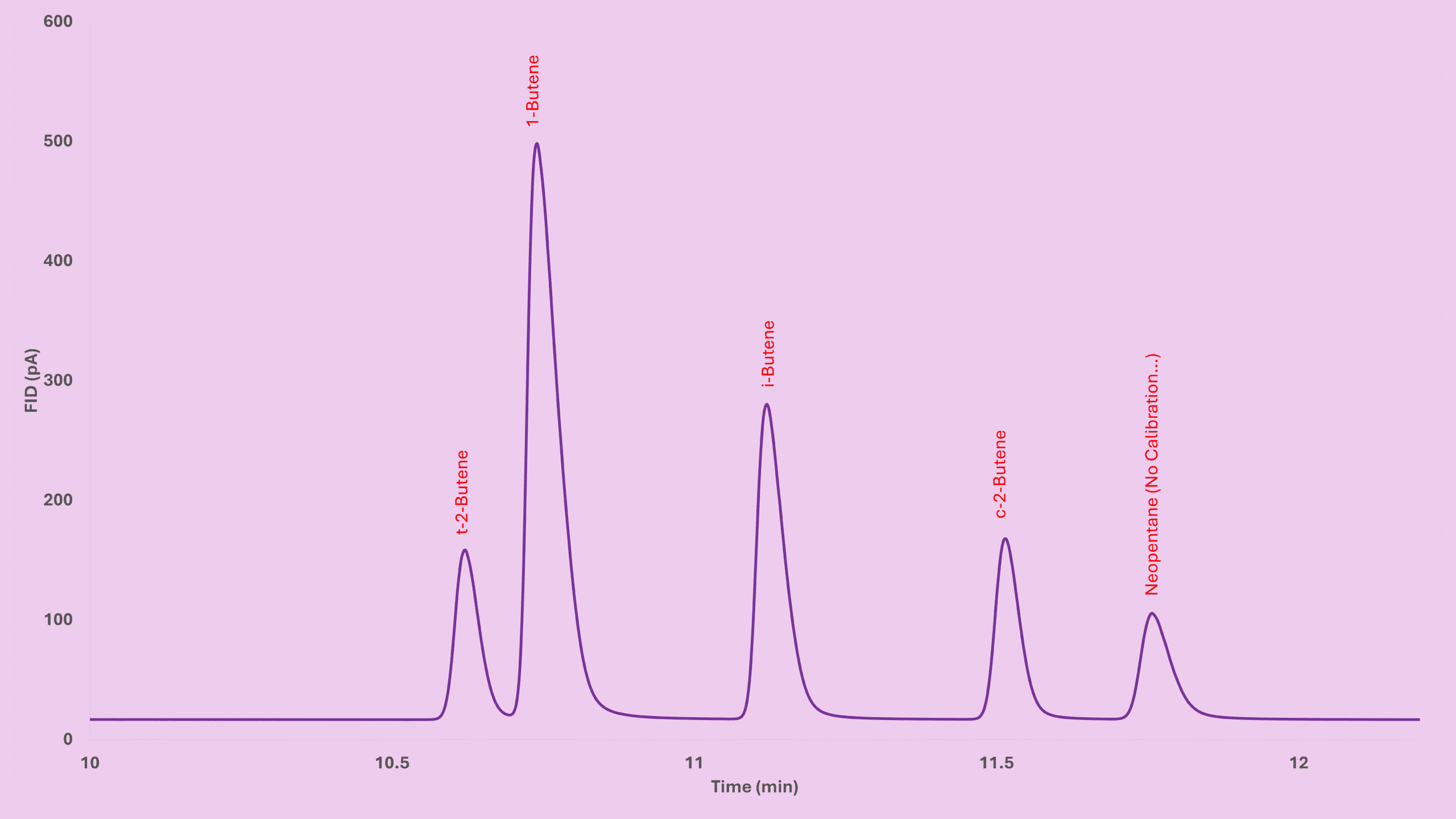
This part is always fun — comparing the calculated values with real experimental data. Here’s one dataset obtained from a standard mixture with known concentrations. Look closely at the last column to see how well theory matches reality.
Experimental vs Theoretical RRF (Mass-Based)
| Component | Mass% | Peak Area | RF | RRF (By Mass, n-Butane Ref) | Carbon | MW | Theoretical RRF (By Mass) | RRF Difference (%) |
|---|---|---|---|---|---|---|---|---|
| Methane | 0.90 | 927.66 | 0.00096937 | 1.135 | 1 | 16 | 1.103 | 2.84% |
| Ethane | 0.05 | 60.09 | 0.00089194 | 1.044 | 2 | 30 | 1.034 | 0.93% |
| Propylene | 0.08 | 93.01 | 0.00081667 | 0.956 | 3 | 42 | 0.966 | -0.99% |
| n-Butane | 6.25 | 7315.81 | 0.00085426 | 1.000 | 4 | 58 | 1.000 | 0.00% |
| 1-Butene | 15.42 | 18426.80 | 0.00083676 | 0.980 | 4 | 56 | 0.966 | 1.45% |
| t-2-Butene | 16.52 | 19409.80 | 0.00085124 | 0.996 | 4 | 56 | 0.966 | 3.21% |
| 1,2-Butadiene | 0.19 | 252.47 | 0.00074428 | 0.871 | 4 | 54 | 0.931 | -6.42% |
| Isopentane | 0.65 | 770.12 | 0.00084898 | 0.994 | 5 | 72 | 0.993 | 0.07% |
| Ethylene | 2.08 | 2426.09 | 0.00085827 | 1.005 | 2 | 28 | 0.966 | 4.06% |
| Propane | 0.16 | 183.84 | 0.00085102 | 0.996 | 3 | 44 | 1.011 | -1.51% |
| Isobutane | 21.01 | 24344.90 | 0.00086316 | 1.010 | 4 | 58 | 1.000 | 1.04% |
| Isobutene | 22.90 | 26878.50 | 0.00085184 | 0.997 | 4 | 56 | 0.966 | 3.28% |
| c-2-Butene | 12.24 | 14504.40 | 0.00084404 | 0.988 | 4 | 56 | 0.966 | 2.33% |
| 1,3-Butadiene | 0.59 | 716.26 | 0.00081897 | 0.959 | 4 | 54 | 0.931 | 2.97% |
| n-Pentane | 0.65 | 807.98 | 0.00080919 | 0.947 | 5 | 72 | 0.993 | -4.62% |
| n-Hexane | 0.31 | 349.21 | 0.00087962 | 1.030 | 6 | 86 | 0.989 | 4.17% |
And just like before — the match is surprisingly close.
Before we wrap up, here’s a gentle reminder: everything we derived above is based on defining the response factor as RF = C / A. That’s why the final formula looks like this:
RRF(i/ref) = (C_ref * MW_i) / (C_i * MW_ref)
If you prefer using the opposite definition, RF = A / C, then the formula simply flips upside down:
RRF(i/ref) = (C_i * MW_ref) / (C_ref * MW_i)
And that’s it for today. I hope you enjoyed this post as much as I enjoyed putting it together.
Key Takeaways
- The FID mass response factor explains how RF changes when concentrations are mass-based.
- Molecular weight becomes part of the calculation, but the carbon-based relationship still holds.
- Most hydrocarbons have very similar RRF-mass values, which explains why many ASTM methods allow using a single response factor.
- Experimental data matches theory closely, giving confidence in applying this logic.
Try the Instant Calculator to Calculate FID mass response factor
A calculator that applies the formulas discussed in this blog post can be found at FID Relative Response Factor Calculator for GAS or FID Relative Response Factor Calculator for LIQUID. Just enter:
- Number of carbon atoms and hydrogen atoms for a reference compound
- Number of carbon atoms and hydrogen atoms for an unknown compound
The result updates automatically — perfect for method development and troubleshooting.
Try This Next
Curious how the same logic works for volume-based concentrations?
Visit the Flame Ionization Detector (FID) to follow the next article in this response-factor series.
