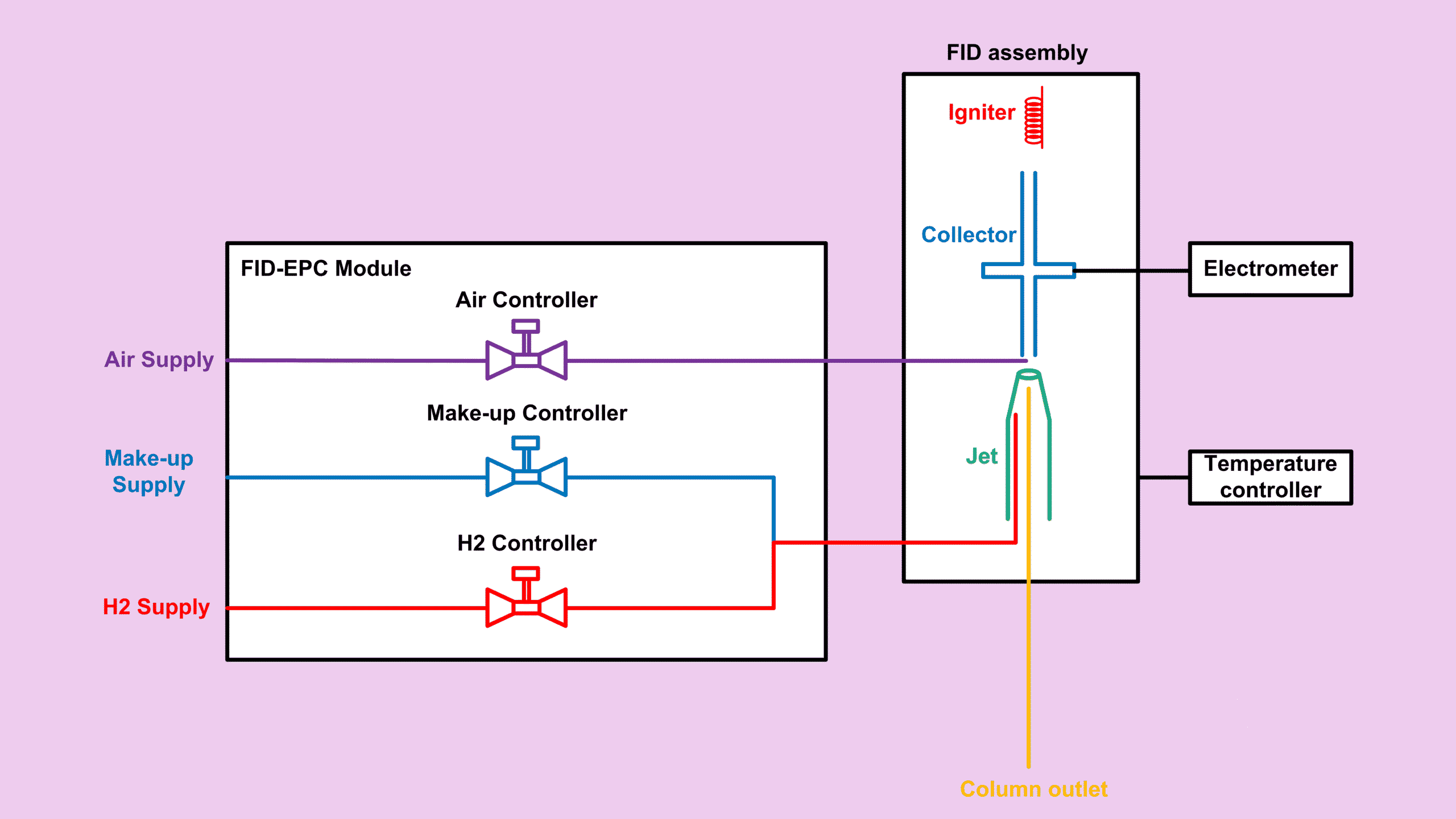
If you ever find yourself hunting through old notebooks just to recall a simple FID relative response factor formula, this post will save your day. I’ve gathered everything into one tidy summary so you can apply the formulas instantly, whether you’re working with gas or liquid hydrocarbons. And because the two calculators sit right at the top of this page, you can test any compound right away without doing the math manually.
FID RRF Calculator for Gas
FID RRF Calculator for Liquid
Summary of FID Relative Response Factor Formulas
Before we dive into the equations, here’s a quick reminder. These formulas work only for compounds that contain carbon and hydrogen (CxHy). They don’t apply to molecules with heteroatoms like O, S, or Cl because those elements affect ionization in the flame and break the assumptions used in these relationships. Now that we’ve cleared that up, let’s walk through each formula.
FID Relative Response Factor by Mole
Because FID responses strongly correlate with the number of carbon atoms in a hydrocarbon, the mole-based RRF is beautifully simple:
RRF_Mole (i/ref) = C_ref / C_i
FID Relative Response Factor by Mass
When you quantify using mass instead of mole fraction, you include molecular weights:
RRF_mass (i/ref) = (C_ref * MW_i) / (C_i * MW_ref)
FID Relative Response Factor by Volume (Ideal Gas)
If you’re working with ideal gas behavior, it stays straightforward:
RRF_Vol (i/ref) = C_ref / C_i
FID Relative Response Factor by Volume (Real Gas)
Real gases introduce compressibility (Z), so the equation adjusts:
RRF_Vol (i/ref) = (C_ref * Z_i) / (C_i * Z_ref)
FID Relative Response Factor by Volume (Liquid)
For liquid calculations, you include density in the relationship:
RRF_Vol (i/ref) = (C_ref * D_ref * MW_i) / (C_i * D_i * MW_ref)
What Each Symbol Means
To keep things easy to recall:
- RRF → relative response factor
- i → compound of interest
- ref → reference compound
- C → number of carbon atoms
- MW → molecular weight
- Z → compressibility factor
- D → relative density or density

A Quick but Essential Reminder
All these relationships assume the definition:
RF = C / A
If you’re using the inverse definition (RF = A / C), then every RRF formula above simply flips upside down. It’s an easy adjustment but important to keep in mind when switching between labs or methods.
Want to Try These Formulas Instantly?
Right at the top of this post, you’ll find a calculator for gas and another for liquid hydrocarbons. I kept them simple with one reference compound and two unknowns. You can use them as often as you want.
If you prefer something more advanced — including a built-in database for MW, carbon count, density, and Z values, plus the ability to calculate multiple components at once — check out my Premium GC Templates linked inside the blog.
Key Takeaway
You now have a compact toolbox of FID relative response factor formulas for gases and liquids. These equations make GC quantification faster and more consistent, especially when you need a quick reference without flipping through large manuals.
Try Other Free GC Calculators
If you found this helpful, feel free to explore more Free GC Calculators and summaries available on the website. They’re all designed to simplify your daily workflow and give you confidence in every calculation.
If you need a refresher on my FID RRF series, check out the links below: