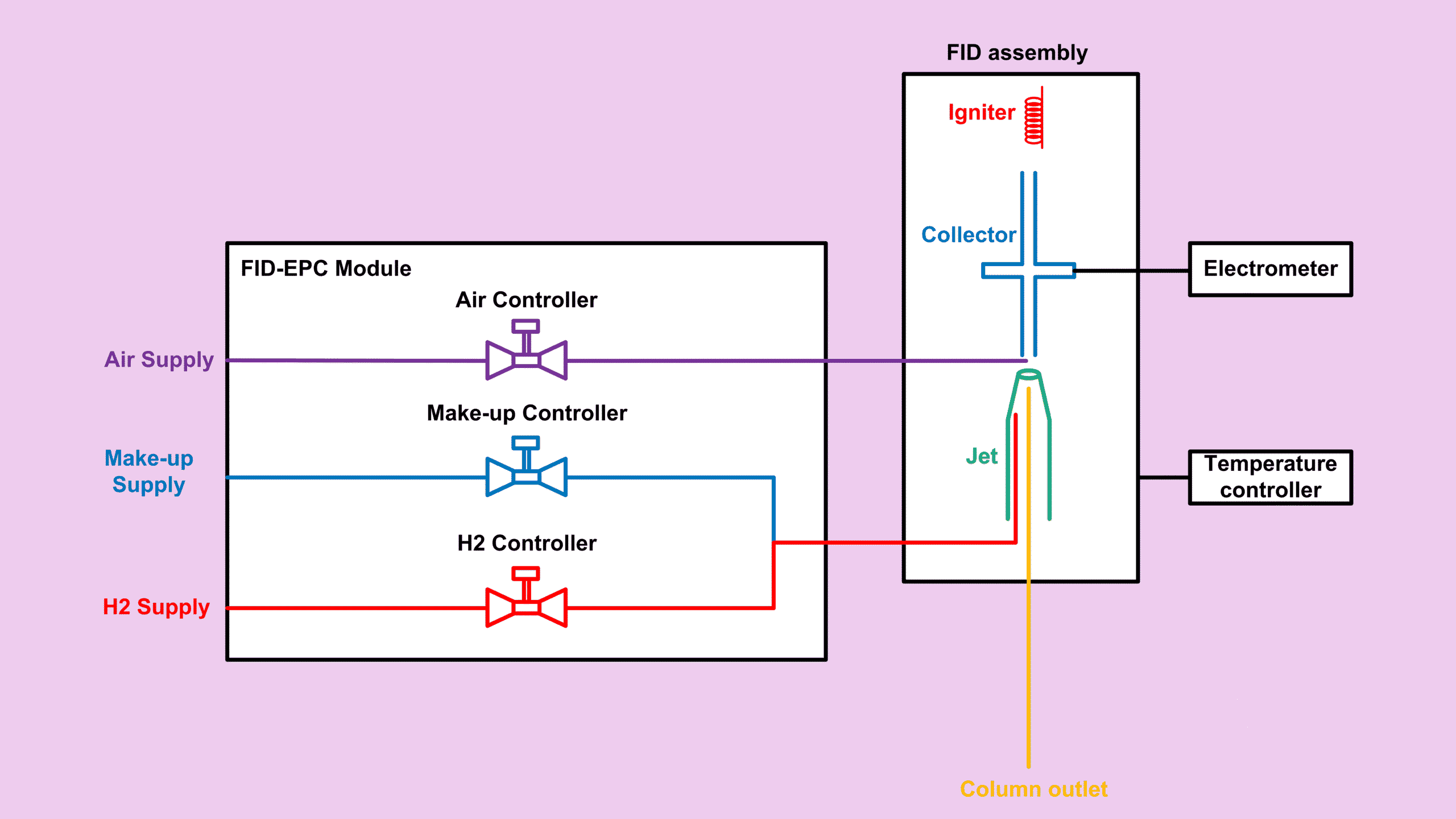
I believe by now you’re already familiar with how to derive a general formula for the relative response factor for hydrocarbons. If not, I strongly recommend revisiting my last two posts in the FID series.
- Understanding the FID Mole Response Factor (A Practical Walkthrough)
- Understanding the FID Mass Response Factor (A Clear and Friendly Walkthrough)
Once you master those concepts, your understanding moves to another level.
In this post, we’ll continue that journey by working through the FID volume response factor, which is the last piece of the RRF puzzle after mole-based and mass-based calculations. To be honest, I was a little hesitant to write this one, simply because volume isn’t a common unit in gas chromatography. But since we covered mole and mass, it wouldn’t feel fair to leave out volume.
How to Think About the FID Volume Response Factor
When it comes to mole or mass, things are quite straightforward whether your components are gases or liquids. However, volume is trickier because the conversion method depends heavily on whether you’re working with a gas or a liquid.
The idea behind handling volume is still similar to handling mass:
convert the volume unit into a mole-related unit first, then derive the response factor.
But because gases and liquids behave differently, their conversions are different too.
Let’s start with the easier one: gases.
FID Volume Response Factor for Gases
To convert gas volume to a mole-related unit, we use the gas law:
P * V = z * n * R * T
I won’t go deep into the equation since it’s not the focus of this post. Most of the time, we assume gases behave as ideal gases where z = 1. Under this assumption, volume (V) becomes directly proportional to moles (n).
Meaning:
- A mixture with 1 mole% Methane and 3 mole% Propane
≈
1 vol% Methane and 3 vol% Propane
Therefore, we can simply apply the mole-based formula from the earlier post:
RRF_Mole(i/ref) = C_ref / C_i
Rewriting it for volume:
RRF_Vol(i/ref) = C_ref / C_i
Where:
- Cᵢ = carbon number of the compound of interest
- C_ref = carbon number of the reference compound
This ideal gas assumption works beautifully for most cases. However, if you want higher accuracy, you can include the compressibility factor (z). Compressibility is both temperature- and pressure-dependent, and a great resource to obtain z-values is ISO 6976 (2025 edition).
Here’s a partial reference table from ISO 6976:
| Component | z @ 0°C, 101.325 kPa | z @ 0°C, 101.325 kPa | z @ 0°C, 101.325 kPa |
|---|---|---|---|
| Methane | 0.9976 | 0.9980 | 0.9981 |
| Ethane | 0.9900 | 0.9915 | 0.9920 |
| Propane | 0.9789 | 0.9821 | 0.9834 |
| n-Butane | 0.9572 | 0.9650 | 0.9682 |
| n-Pentane | 0.9180 | 0.9370 | 0.9450 |
| n-Hexane | 0.8920 | 0.9130 | 0.9190 |
Since z differs by gas, to get moles from volume, we rearrange the gas law:
V / z → mole-related volume
Now, let’s go back to our FID flame reaction:
CxHy + O2 → xCHO+ + e−
Take Methane and Propane as examples. Assume we have a gas mixture with “x” vol% Methane and “x” vol% Propane.
- “x” vol% Methane → x / z_Methane mole-related
- “x” vol% Propane → x / z_Propane mole-related
Now that both are mole-related, we can reuse the approach from previous posts:
x/z_Methane of Methane_Mole → "y" FID peak area
So for the other:
x/z_Propane of Methane_Mole → (x/z_Propane * y) / (x/z_Methane)
Thus Propane, which produces 3 CHO⁺ ions per molecule, gives:
x/z_Propane of Propane_Mole → 3 * (x/z_Propane * y) / (x/z_Methane)
Now we calculate their response factors:
RF_Vol(Methane) = x / y
RF_Vol(Propane) = x / (3 * (z_Methane * y) / z_Propane)
Relative response factor (Methane as reference):
RRF_Vol(Propane/Methane)
= (x / (3*(z_Methane*y)/z_Propane)) / (x/y)
Solving:
RRF_Vol(Propane/Methane) = 1 / (3 * z_Methane / z_Propane)
Or rewritten:
RRF_Vol(Propane/Methane) = z_Propane / (3 * z_Methane)
Since the number 3 represents the carbon ratio between Propane and Methane, we can generalize:
RRF_Vol(i/ref) = z_i / ((C_i / C_ref) * z_ref)
Or:
RRF_Vol(i/ref) = (C_ref * z_i) / (C_i * z_ref)
To summarize the gas phase:
If ideal gas:
RRF_Vol(i/ref) = C_ref / C_i
If real gas:
RRF_Vol(i/ref) = (C_ref * z_i) / (C_i * z_ref)
FID Volume Response Factor for Liquids
For liquids, we must convert volume to mass first using density (d), then from mass to moles using molecular weight (MW). Density also depends on temperature and pressure, so make sure you use the correct value.
Assume a liquid mixture containing “x” vol% Benzene and “x” vol% Toluene.
Conversion steps:
"x" vol% Benzene → x * d_Benzene (mass-related)
"x" vol% Toluene → x * d_Toluene (mass-related)
Then convert mass to moles:
(x * d_Benzene) / MW_Benzene
(x * d_Toluene) / MW_Toluene
Now for their FID peak areas:
(x*d_Benzene)/MW_Benzene of Benzene_Mole → "y" peak area
So:
(x*d_Toluene)/MW_Toluene of Benzene_Mole
→ ((x*d_Toluene)/MW_Toluene) / ((x*d_Benzene)/MW_Benzene) * y
Since Toluene has 7 carbons and Benzene has 6:
(x*d_Toluene)/MW_Toluene of Toluene_Mole → 7/6 * ((x*d_Toluene)/MW_Toluene) / ((x*d_Benzene)/MW_Benzene) * y
Response factors:
RF_Vol(Benzene) = x / y
RF_Vol(Toluene) = x / (7/6 * ((x*d_Toluene)/MW_Toluene)/((x*d_Benzene)/MW_Benzene) * y)
Relative response factor:
RRF_Vol(Toluene/Benzene)
= (x/(7/6*((x*d_Toluene)/MW_Toluene)/((x*d_Benzene)/MW_Benzene)*y)) / (x/y)
Solving:
RRF_Vol(Toluene/Benzene)
= 1 / (7/6 * (d_Toluene/MW_Toluene) / (d_Benzene/MW_Benzene))
Or:
RRF_Vol(Toluene/Benzene)
= 1 / (7/6 * (d_Toluene * MW_Benzene) / (d_Benzene * MW_Toluene))
Finally:
RRF_Vol(Toluene/Benzene)
= (6 * d_Benzene * MW_Toluene) / (7 * d_Toluene * MW_Benzene)
Generalized:
RRF_Vol(i/ref) = (C_ref * d_ref * MW_i) / (C_i * d_i * MW_ref)
Before we wrap up, here’s a quick reminder: everything in this post is based on defining the response factor as RF = C / A. That’s why all the FID volume response factor formulas above follow that structure. If you prefer using the alternative definition, RF = A / C, then you simply flip each RRF formula upside down. The logic stays the same — just the math inverts.
Try the Instant Calculator
A calculator that applies the formulas discussed in this blog post can be found at FID Relative Response Factor Calculator for GAS or FID Relative Response Factor Calculator for LIQUID. Just enter:
- Number of carbon atoms, hydrogen atoms and compressibility factor/density for a reference compound
- Number of carbon atoms, hydrogen atoms and compressibility factor/density for an unknown compound
The result updates automatically, perfect for method development and troubleshooting.
Key Takeaways
- The FID volume response factor works for both gases and liquids, but each requires a different conversion path.
- For gases, volume converts to moles through compressibility or ideal gas assumptions.
- For liquids, volume must first convert to mass via density, then to moles through MW.
- The final RRF formulas match the logic from mole- and mass-based calculations.
- Understanding this completes the trilogy of mole → mass → volume RRF methods.
Your Next Step
If you enjoyed this post, follow the next article where I’ll summarize all three RRF concepts — mole, mass, and volume — and provide a handy calculator to compute RRF instantly.
Meanwhile, visit Free GC Calculators for a handful of calculators.