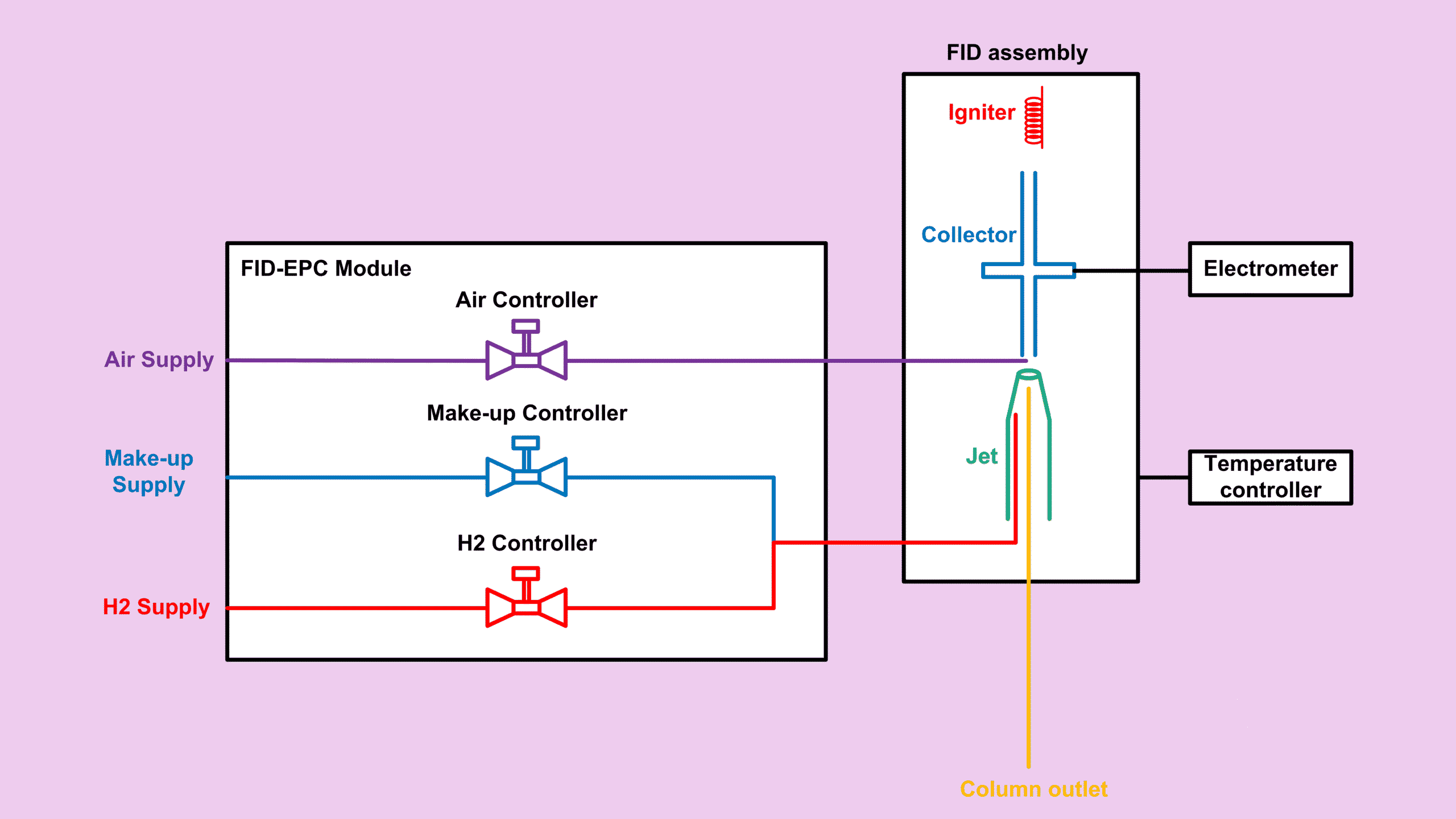
If you’ve been following this FID series, you already know why RRF matters. In this post, we’ll focus on FID RRF Verification, specifically how to compare theoretical relative response factors with experimental relative response factors in a practical and straightforward way.
Previously, we discussed how RRF helps quantify unknown compounds. Now, however, we move one step further. FID RRF Verification allows you to confirm whether your GC system behaves as expected and whether theoretical values are still valid for your setup.
Before we dive in, just to let you know that this article is part of my Flame Ionization Detector (FID) series which you can find the summary for all other posts related to FID at FID Detector: Complete Guide to the Entire FID Series.
Why FID RRF Verification Matters
Some international standard methods allow the use of theoretical RRF values instead of determining them experimentally every time. One example is UOP Method 744.
Method 744 focuses on quantifying aromatic content in hydrocarbon samples. Importantly, it provides a table of theoretical mass-based RRF values, as below:
| Name | RRF_Mass |
|---|---|
| n-Heptane | 1.000 |
| Benzene | 0.909 |
| Toluene | 0.919 |
| Xylenes | 0.927 |
| 1-Methyl-2-ethylbenzene | 0.933 |
| 1,2,3,4-Tetramethylbenzene | 0.938 |
Notice that n-Heptane has RRF = 1. That means it is the reference compound.
If you’re unsure where these mass-based values come from, you can revisit my earlier post:
➡ Understanding the FID Mass Response Factor (A Clear and Friendly Walkthrough)
However, there is a condition.
Method 744 allows theoretical RRF values only if they are within 5% of experimental values. Therefore, you still need to inject a known mixture at least once to confirm system performance. After major maintenance or hardware changes, repeating this FID RRF Verification becomes necessary.
How to Perform FID RRF Verification Step by Step
First, remember that RRF is a ratio between the response factor of an interested compound and a reference compound.
So, after injecting a known mixture, you will have:
- Concentration of each compound
- Peak area of each compound
From there, calculate response factors:
\[ RF_{ref} = \frac{C_{ref}}{A_{ref}} \] \[ RF_{i} = \frac{C_{i}}{A_{i}} \]Then calculate the experimental RRF:
\[ RRF_{i} = \frac{RF_{i}}{RF_{ref}} \]Next, obtain the theoretical RRF. You can calculate it using the FID RRF Calculator for Liquid – Relative Response Factor Calculator on this site, or retrieve it directly from published standards such as UOP 744.
Finally, calculate the relative difference:
\[ {Relative Difference (\%)} = \frac{RRF_{experimental} – RRF_{theoretical}}{RRF_{theoretical}} \times 100 \]That’s the core equation behind FID RRF Verification.
If the result stays within ±5% as per UOP744 your theoretical RRF remains acceptable. In some other methods, the acceptable difference could be up to 10%.
Practical Example of FID RRF Verification Using UOP 744 Data
Let’s walk through real numbers.
| Name | Mass% | Area |
|---|---|---|
| n-Heptane | 24.763 | 19168.47 |
| Benzene | 14.936 | 12682.40 |
| Toluene | 19.958 | 16678.67 |
| Xylenes | 20.014 | 16625.97 |
| 1-Methyl-2-ethylbenzene | 9.923 | 8184.23 |
| 1,2,3,4-Tetramethylbenzene | 10.041 | 8257.82 |
For demonstration purposes, we will verify Benzene and Toluene only. You can repeat the same workflow for the remaining compounds.
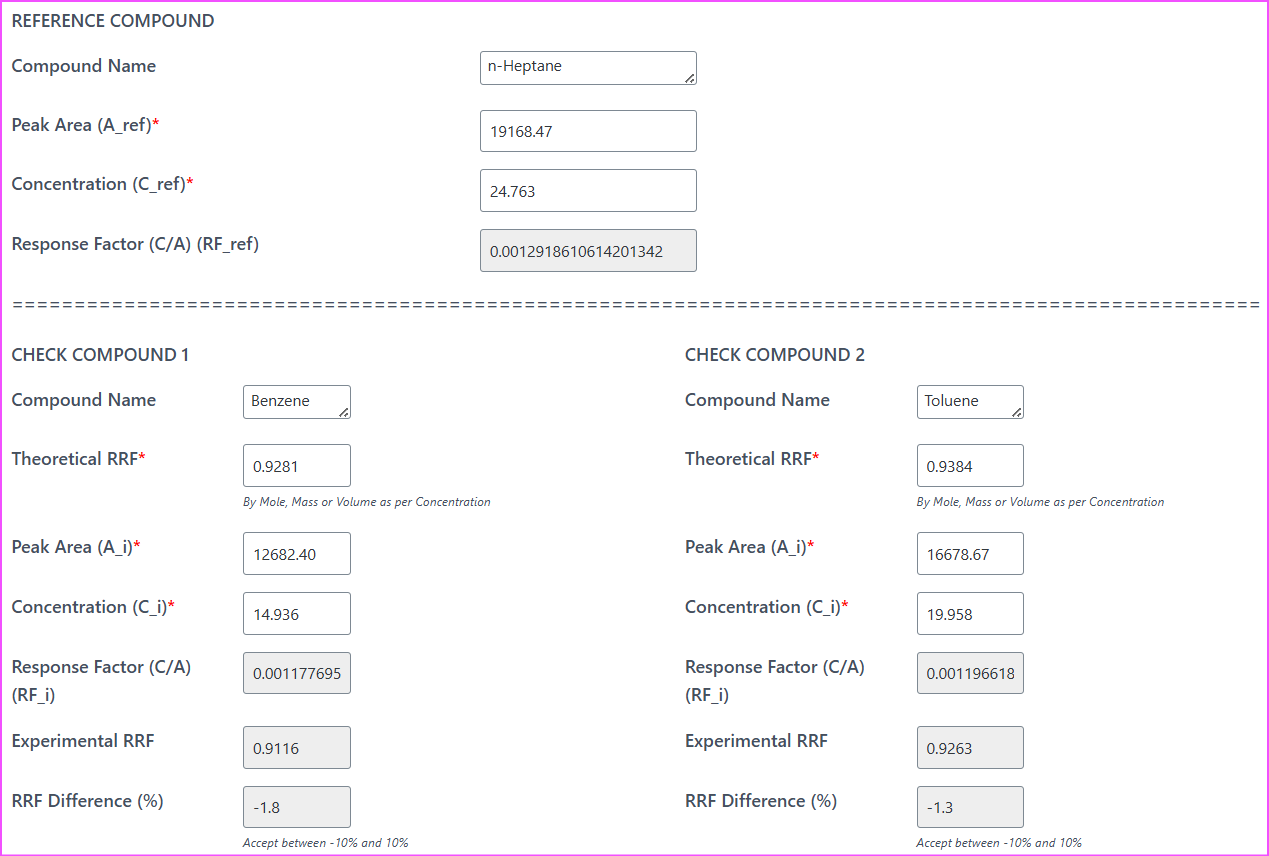
Instead of building spreadsheets manually, you can simply use the FID RRF Verification Calculator (Theoretical vs Experimental) on this site.
Enter:
- Reference compound (n-Heptane)
- Concentration values
- Peak areas
- Theoretical RRF values
Within seconds, the calculator tells you whether your system meets the 5% criterion.
This makes the verification fast, repeatable, and far less error-prone.
Key Takeaway
FID RRF Verification is not complicated. In fact, it involves only:
- Calculating response factors
- Determining experimental RRF
- Comparing against theoretical RRF
- Checking the percentage difference
However, this simple verification step ensures:
- Compliance with methods like UOP 744
- Confidence in quantitative results
- Reduced need for frequent recalibration
Most importantly, it gives you clarity about how your FID truly performs.
Continue Exploring
If this helped clarify how to FID RRF Verification works, explore the other related posts below: