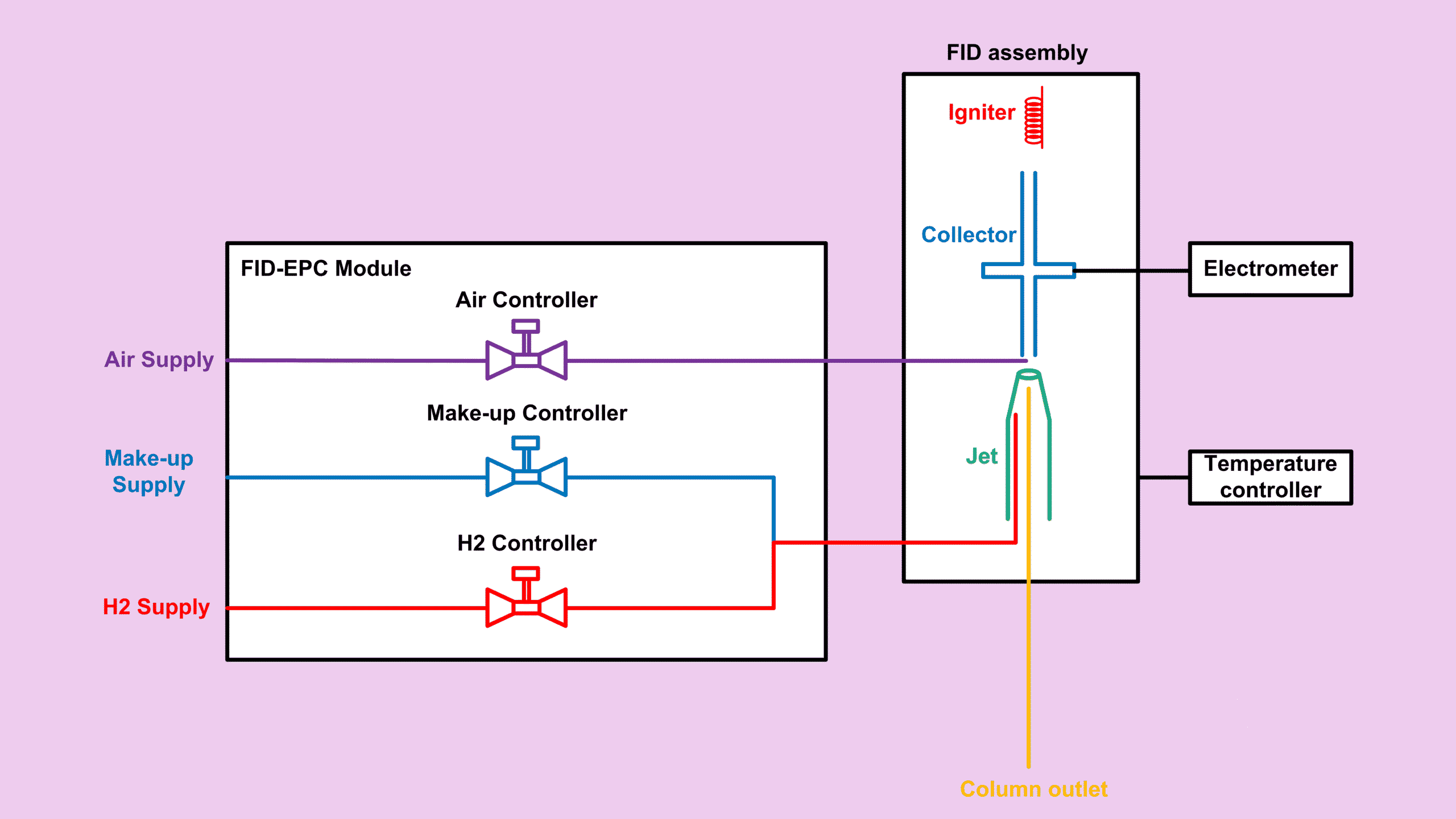
If you’ve read my earlier posts about FID, you already know that FID CO CO2 Detection is not naturally possible. A FID does not respond well to compounds without a C–H bond, such as CO and CO₂. However, with the help of a methanizer, the detection becomes practical and reliable.
So how does this actually work?
Before we dive in, just to let you know that this article is part of my Flame Ionization Detector (FID) series which you can find the summary for all other posts related to FID at FID Detector: Complete Guide to the Entire FID Series.
What Makes FID CO CO2 Detection Possible?
The key is methane.
A methanizer is an add-on device that converts CO and CO₂ into methane (CH₄), which a normal FID can easily detect.
Here are the reactions involved:
CO + 3H2 → CH4 + H2O
CO2 + 4H2 → CH4 + 2H2O
Because methane contains C–H bonds, the FID responds strongly to it. Therefore, when we talk about FID CO CO2 Detection, what we truly mean is that the FID is detecting methane converted from CO and CO₂.
The reaction requires:
- Hydrogen (H₂)
- High temperature (375–450 °C)
- Nickel catalyst
Below is a simple diagram to visualize how a methanizer looks.
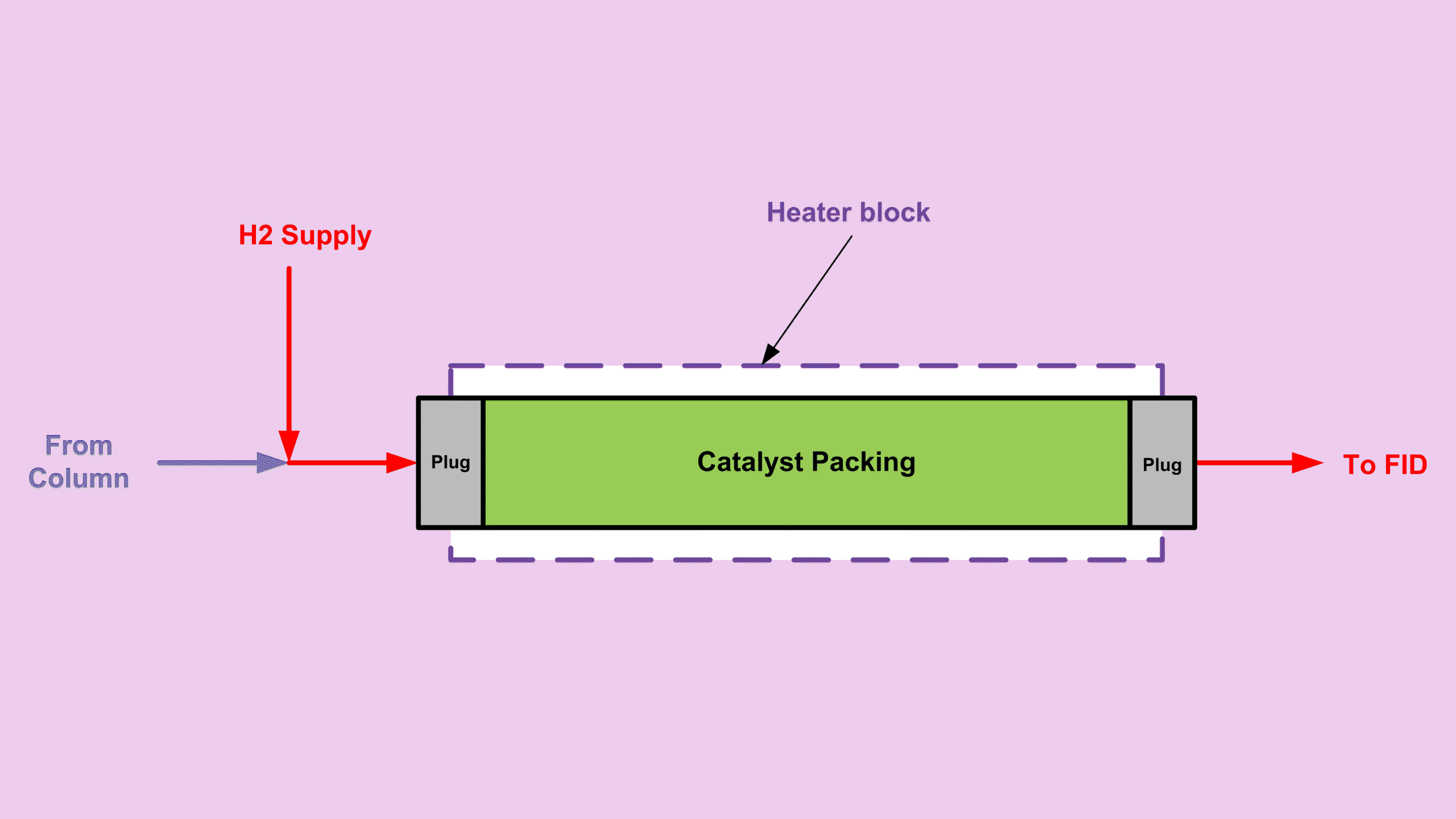
Conversion Efficiency in FID CO CO2 Detection
At this point, a logical question arises:
If the FID only detects methane, how do we know methane truly represents all the CO and CO₂?
The answer is conversion efficiency (CE).
Ideally, we want 100% conversion. In practice, ≥90% is generally acceptable. However, standards like UOP603 require the response factor difference between CH₄ and CO/CO₂ to be within 5%, which corresponds roughly to a minimum of 95% conversion efficiency.
Deriving Conversion Efficiency (Mole Basis)
Let’s assume:
- 10 ppm mol CO
- 11 ppm mol CH₄
- 12 ppm mol CO₂
- CH₄ area = 100
From proportionality, 10 ppm CH4 gives:
\[ \frac{10 \times 100}{11} = 90.9 \]Therefore, if CO converts 100%, it should produce 90.9 area counts.
If measured area = 89:
\[ CE = \frac{89}{90.9} \times 100\% = 98\% \]Generalized Formula (Mole Basis)
Replace the area and concentration in the example above:
\[ CE_{(Mole/CO)} = \frac{Area_{CO}}{\frac{Mole_{CO} \times {Area_{CH4}}}{Mole_{CH4}}} \times 100\% \]Rearrange the formula:
\[ CE_{(Mole/CO)} = \frac{\frac{Mole_{CH4}}{Area_{CH4}}}{\frac{Mole_{CO}}{Area_{CO}}} \times 100\% \]Replace RF = C / A:
\[ CE_{(Mole/CO)} = \frac{RF_{(Mole/CH4)}}{RF_{(Mole/CO)}} \times 100\% \] \[ CE_{(Mole/CO2)} = \frac{RF_{(Mole/CH4)}}{RF_{(Mole/CO2)}} \times 100\% \]Because ppm mol and ppm vol behave similarly under near-ideal gas conditions, the same logic applies for gas standards expressed in volume basis.
👉 Refer to my earlier post on Relative Response Factor (RRF) for a refresher on RF calculations.
- Understanding the FID Mole Response Factor (A Practical Walkthrough)
- Understanding the FID Mass Response Factor (A Clear and Friendly Walkthrough)
- Understanding the FID Volume Response Factor (A Friendly Deep Dive)
Deriving Conversion Efficiency (Mass Basis)
When working in ppm mass, molecular weight must be considered.
Example:
- CH₄ MW = 16
- CO MW = 28
- CO₂ MW = 44
Generalized Formula (Mass Basis)
\[ CE_{(Mole/CO)} = \frac{RF_{(Mass/CH4)}}{RF_{(Mass/CO)}} \times \frac{MW_{CO}}{MW_{CH4}} \times 100\% \] \[ CE_{(Mole/CO2)} = \frac{RF_{(Mass/CH4)}}{RF_{(Mass/CO2)}} \times \frac{MW_{CO2}}{MW_{CH4}} \times 100\% \]Even if methane is not your analyte of interest, your calibration gas must contain CH₄ for proper FID CO CO2 Detection verification.
Moreover, CE changes over time. Therefore, always check it after recalibration or maintenance.
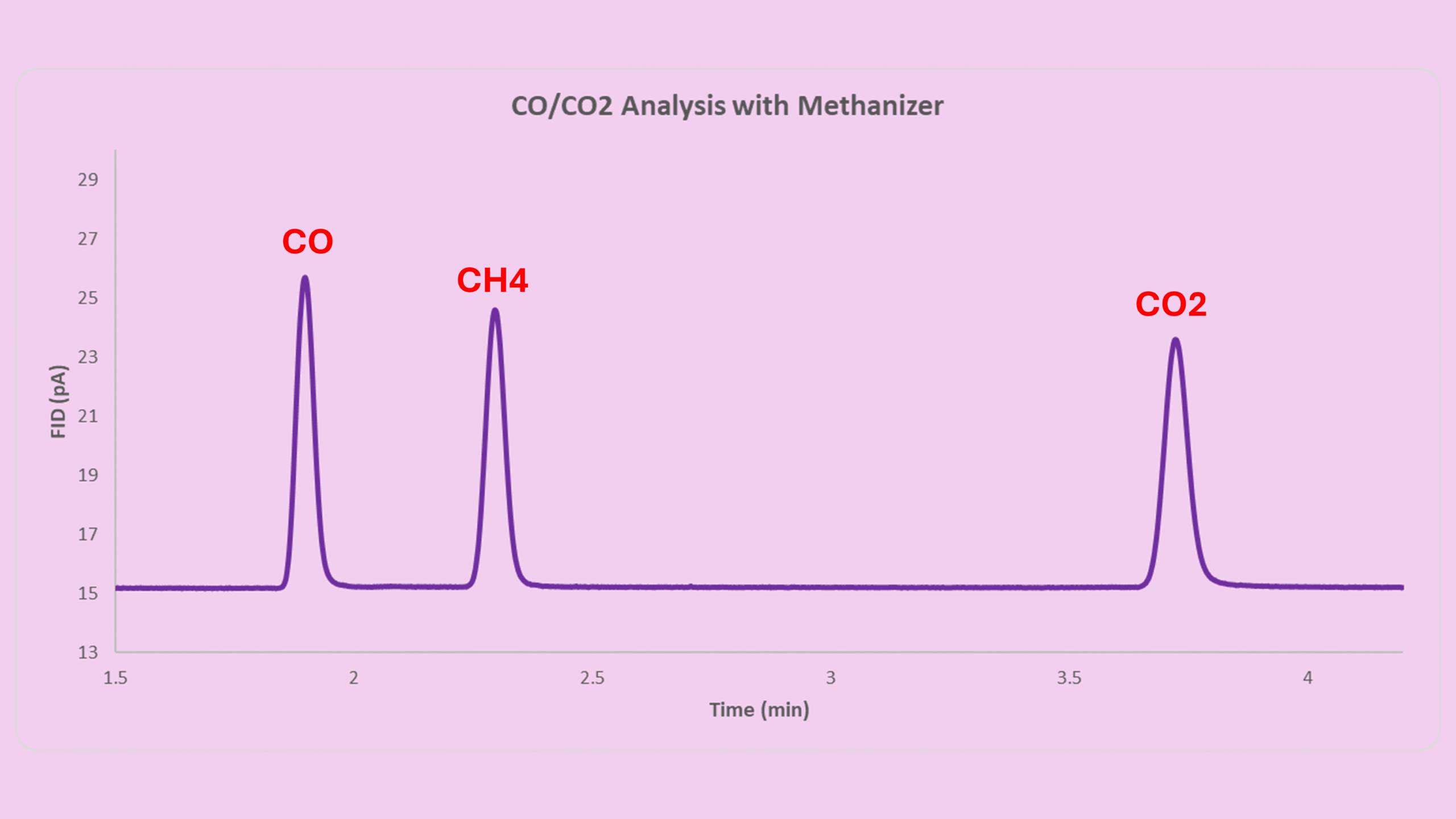
Real Data Example for UOP603 Compliance
Here is real calibration data used for FID CO CO2 Detection:
| Component | Certified (ppm mol) | Peak Area | RF | RRF Relative to CH4 | Difference % |
|---|---|---|---|---|---|
| Methane | 25.10 | 134.77399 | 0.186238 | 1.000 | 0.00% |
| Carbon Monoxide | 5.02 | 26.40539 | 0.190113 | 1.021 | 2.08% |
| Carbon Dioxide | 5.02 | 28.34049 | 0.177132 | 0.951 | 4.89% |
Both CO and CO₂ meet the ≤5% difference requirement.
Interestingly, I sometimes use this calculation to verify whether the standard cylinder itself is reliable, because COA values are not always perfect in real industrial settings.
Practical Issues in FID CO CO2 Detection
Although FID CO CO2 Detection works well, there are several practical concerns.
1. Peak Tailing
The conversion reaction can introduce slight peak tailing.
2. Heavy Hydrocarbon Interference
Methanizers strongly retain hydrocarbons.
When large or high-concentration hydrocarbons enter the methanizer:
- Baseline drifts slowly downward
- Heavy compounds stick and elute very slowly
For example, trace CO/CO₂ analysis in propylene bulk requires:
- Proper column separation to separate bulk propane from CO and CO2
- Backflush or diversion hardware to avoid bulk propane from entering the methanizer
I will cover these techniques in a future post.
3. Accidental Heavy HC Contamination
If heavy hydrocarbons enter the methanizer:
- Stop hydrogen
- Replace hydrogen with air
- Maintain 375–450 °C
- Flush for 4–6 hours
- Revert to hydrogen
- Wait 1 hour before re-testing
Air oxidizes retained hydrocarbons into CO₂ and H₂O, effectively cleaning the catalyst.
Key Takeaways
- FID CO CO2 Detection works by converting CO and CO₂ into methane.
- Conversion efficiency determines accuracy.
- CH₄ must be included in calibration gas.
- Heavy hydrocarbons can contaminate the methanizer.
Try the Instant Calculator
A calculator that applies the formulas discussed in this blog post can be found at FID Methanizer RRF Calculator. Just enter:
- Concentration and Peak Area of Methane, Carbon Monoxide and Carbon Dioxide
- Concentration Basis
The result updates automatically, perfect for method development and troubleshooting.
Continue Learning
If you work with trace gas analysis or process GC systems, understanding FID CO CO2 Detection will significantly improve your confidence in calibration and troubleshooting.
Explore the CE calculators above and verify your own system performance.
Feel free to explore other related posts below: